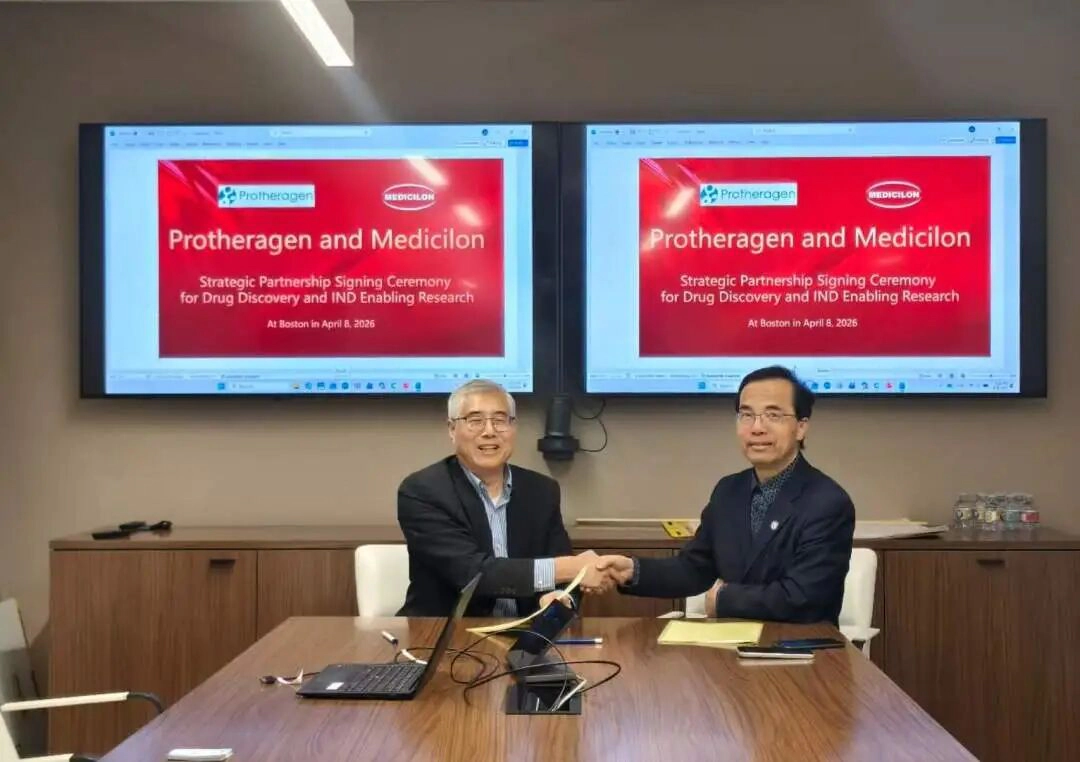
On April 6, Medicilon formally signed a strategic cooperation agreement with Protheragen and its affiliated companies. Leveraging a collaborative “Project + Platform” model, the two parties will integrate Protheragen’s strengths in innovative drug incubation and global project sourcing with Medicilon’s one-stop preclinical pharmaceutical R&D service platform, jointly building an efficient, compliant and internationalized drug R&D ecosystem.

Complementary Strengths for a Win-Win Ecosystem
As an international one-stop preclinical pharmaceutical R&D CRO, Medicilon offers full-process services spanning drug discovery, CMC and preclinical research. It holds certifications compliant with NMPA, FDA, OECD GLP standards and AAALAC, and has built R&D platforms for novel molecular drugs (antibodies, ADCs, mRNA vaccines, etc.). With mature technologies and proven cases, Medicilon has supported the overseas launch of 15+ innovative drugs.

“Project + Platform” Model: Two-Way Empowerment from Idea to IND
This collaboration integrates Protheragen’s global project incubation and cross-border capabilities with Medicilon’s technology platforms and compliant delivery capacity. It will support Protheragen’s global innovative projects with efficient preclinical R&D, while injecting new momentum into Medicilon’s global project sourcing and international layout, jointly building an open, win-win and sustainable global drug R&D ecosystem.
Protheragen Inc.
Headquartered in New York, Protheragen Inc. is an integrated biopharmaceutical incubator dedicated to transforming innovative scientific concepts into high-value commercial assets. Our mission is built on a deep understanding of both the U.S. and dynamic emerging markets, enabling us to help life science companies navigate regional complexities and achieve impactful global strategic partnerships.