Poster I Conjugate Drugs Analysis
- Presenter: Dr. Min Fang
- Time: Wednesday, April 22, 9:00 AM – 12:00 PM
- Location: Poster Section 38, Board #24, Poster #7670
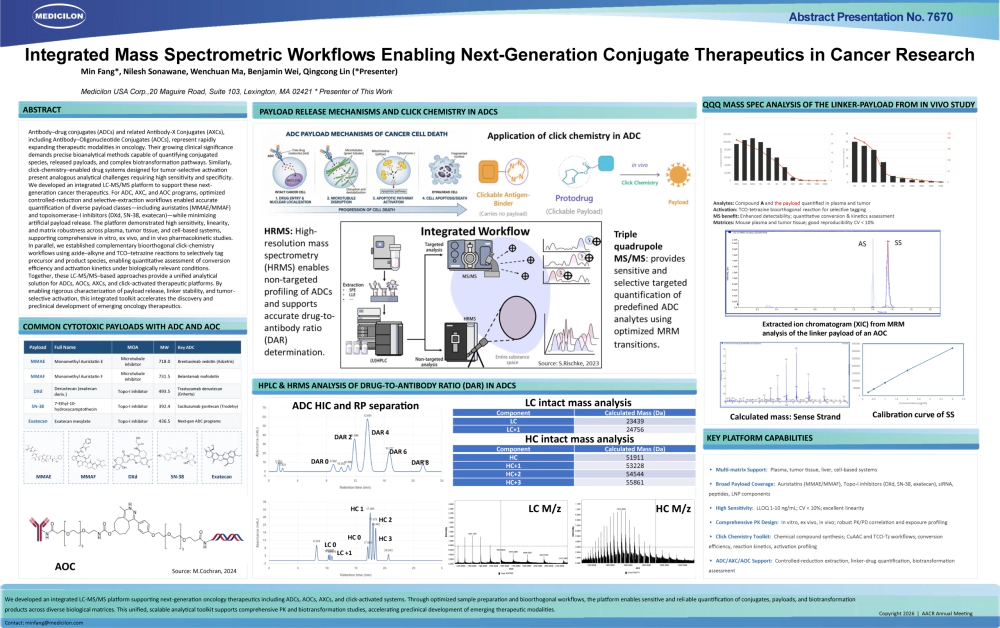
- Title: Integrated Mass Spectrometric Workflows Enabling Next-Generation Conjugate Therapeutics in Cancer Research
- Differentiating total antibody from conjugated antibody: Traditional ligand-binding assays (LBAs) measure total antibody concentration but fail to determine payload attachment, leading to inaccurate drug exposure and stability assessments.
- Detecting free payload and metabolites: Premature payload release or metabolic inactivation affects efficacy and safety, but requires highly sensitive detection methods (often at pg/mL levels).
- Tracking click-chemistry activation: Bioorthogonal activation kinetics and product profiles of prodrugs in the tumor microenvironment are difficult to monitor with routine methods.
Medicilon’s Solution——
Medicilon’s integrated LC-MS/MS workflow features: optimized sample pretreatment to minimize artifactual payload release for accurate in vivo representation; high-sensitivity bioanalysis with validated linearity and matrix tolerance for diverse matrices, enabling microsampling and continuous PK studies; and a dedicated MS-enhanced detection scheme for bioorthogonal click-chemistry, facilitating simultaneous quantification of prodrugs and activated products for PK/PD correlation. This platform reliably supports pharmacokinetic evaluations of ADCs, AOCs, and click-chemistry-activated drugs, providing valuable insights into biotransformation and exposure-response relationships.
Poster II When CAR-T Goes Beyond Oncology
- Presenter: Dr. Ruowen Zhang
- Time: Tuesday, April 21, 2:00 PM – 5:00 PM
- Location: Poster Section 49, Board #25, Poster #6714
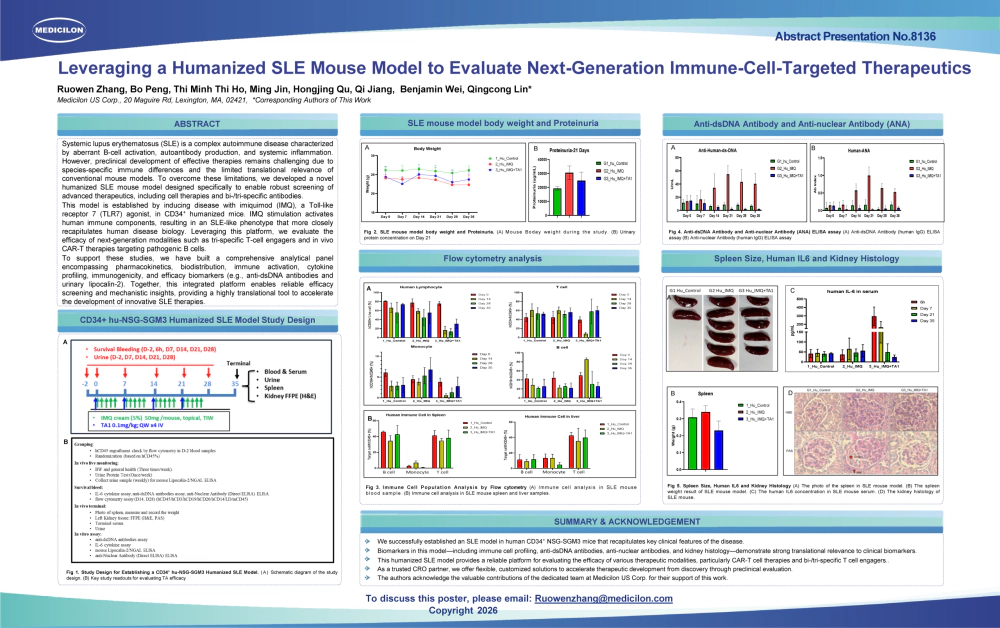
- Title: Humanized SLE Mouse Model for Evaluating B-Cell CAR-T Therapy Efficacy and Safety
- Cytokine release syndrome (CRS): Murine cytokine profiles differ significantly from human profiles, hindering accurate toxicity assessment.
- Anti-drug antibodies (ADA): Human CAR constructs can elicit immunogenic responses in mice, confounding efficacy evaluations.
- Target expression differences: Variations in CD19 expression between humans and mice affect CAR-T expansion and persistence.
Medicilon’s Solution——
Medicilon’s humanized SLE mouse model overcomes preclinical bottlenecks in autoimmune CAR-T therapy through the following key features: a humanized immune system reconstituted with CD34+ hematopoietic stem cells, TLR7 agonist induction using Imiquimod (IMQ) to stimulate human pDCs and B cells, inducing SLE biomarkers mirroring active human lupus, and in vivo CAR-T generation and delivery via anti-CD5 antibody-targeted LNPs encapsulating CD19 CAR DNA, directly transfecting CD5+ T cells in vivo. This in vivo approach bypasses the complexities of ex vivo viral transduction and better reflects future in vivo CAR-T strategies. This model is ideal for assessing CAR-T therapy efficacy, toxicology, and PK/PD in autoimmune disease research.
Poster III AOC from Design to Production
- Presenter: Dr. Ao Huang
- Location: Medicilon Booth #1553
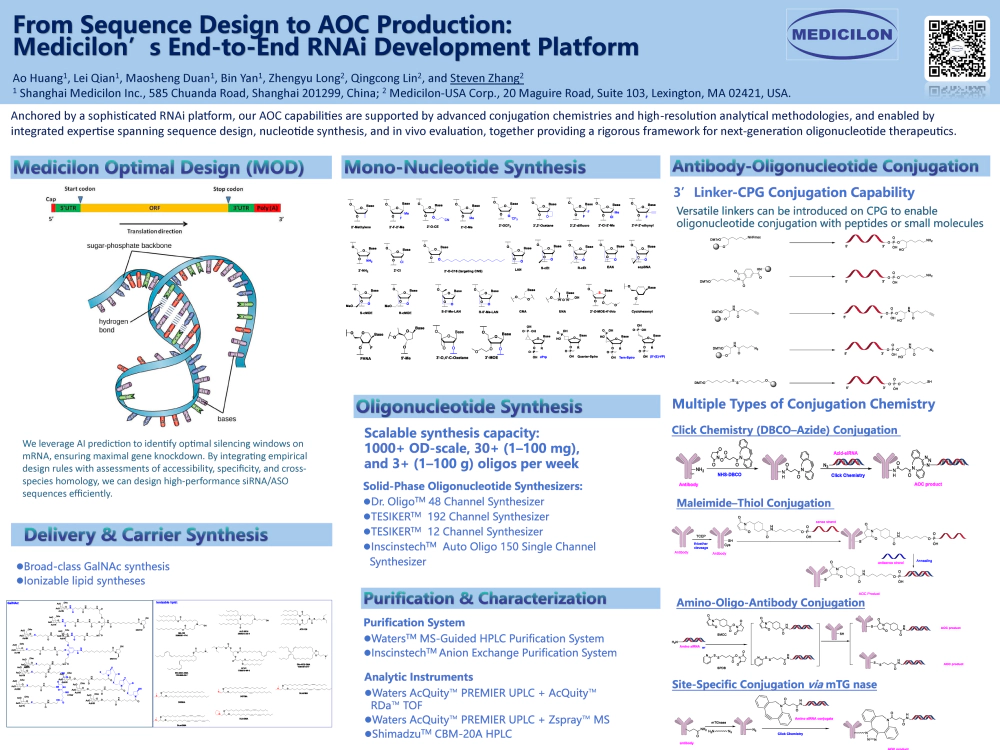
- Title: From Sequence Design to AOC Production: Medicilon’s End-to-End RNAi Development Platform
- Sequence design: Difficulty in accurately identifying optimal mRNA silencing windows, balancing specificity and cross-species homology, leading to low efficiency in siRNA/ASO design.
- Synthesis and conjugation: Insufficient capacity for large-scale oligonucleotide synthesis; lack of diverse, site-specific antibody-oligonucleotide conjugation technologies; difficulty in controlling DAR values.
- Delivery systems: Lack of development capabilities for efficient delivery vectors such as GalNAc and ionizable lipids, hindering efficient delivery of oligonucleotide drugs.
Medicilon’s Solution——
Medicilon’s RNAi-AOC R&D platform provides end-to-end extrahepatic delivery solutions, encompassing sequence design to preclinical evaluation. Our capabilities include: AI-driven sequence design for optimal siRNA/ASO candidate selection; large-scale oligonucleotide synthesis for high-quality payloads; diverse AOC conjugation strategies (click chemistry, maleimide-thiol coupling, AOAC, site-specific conjugation); rigorous AOC purification and characterization (AEX, SEC, UPLC-MS, TOF); and closed-loop development integrating chemical modification, conjugation engineering, analytical characterization, and in vivo DMPK/efficacy evaluation to accelerate AOC discovery.
Beyond Posters Tumor Model Database
- Small-molecule drugs
- Antibodies, ADCs, AOCs
- Cell therapies (CAR-T, TCR-T, CAR-NK)
- Oncolytic viruses, AAV gene therapy
- Nucleic acid drugs (siRNA, ASO)
- And more
Scan the QR code to explore

Top Star Dr. Panda