Recently, MediLink Therapeutics entered into a new exclusive license agreement with Roche (SIX: RO, ROG; OTCQX: RHHBY), a global pharmaceutical giant, for YL201, an antibody-drug conjugate (ADC) targeting B7H3.This landmark deal, featuring an upfront payment and near-term milestone payments exceeding USD 570 million, not only serves as a strong recognition of the substantial clinical value and market potential of the YL201 program, but also stands as a compelling testament to the international competitiveness of MediLink Therapeutics’ proprietary TMALIN® technology platform.
As a long-term partner of MediLink Therapeutics, Medicilion provided efficient Full-Time Equivalent (FTE) services for the YL201 program, facilitating the efficient R&D and optimization of the target molecule for the client, and also participated in the development of MediLink Therapeutics’ TMALIN® technology platform. In addition to completing the synthesis of linkers and payloads for this platform, MediciLion developed a concise and efficient process route for synthesizing payload intermediates from inexpensive and readily available raw materials, laying a solid foundation for the TMALIN platform and the subsequent development of ADC drug candidates.
YL201 Program: A B7H3-Targeting ADC Breaking New Ground in Small Cell Lung Cancer Treatment
The YL201 program licensed to Roche is a key pipeline asset developed on MediLink Therapeutics’ TMALIN® platform, which is an ADC targeting B7H3.
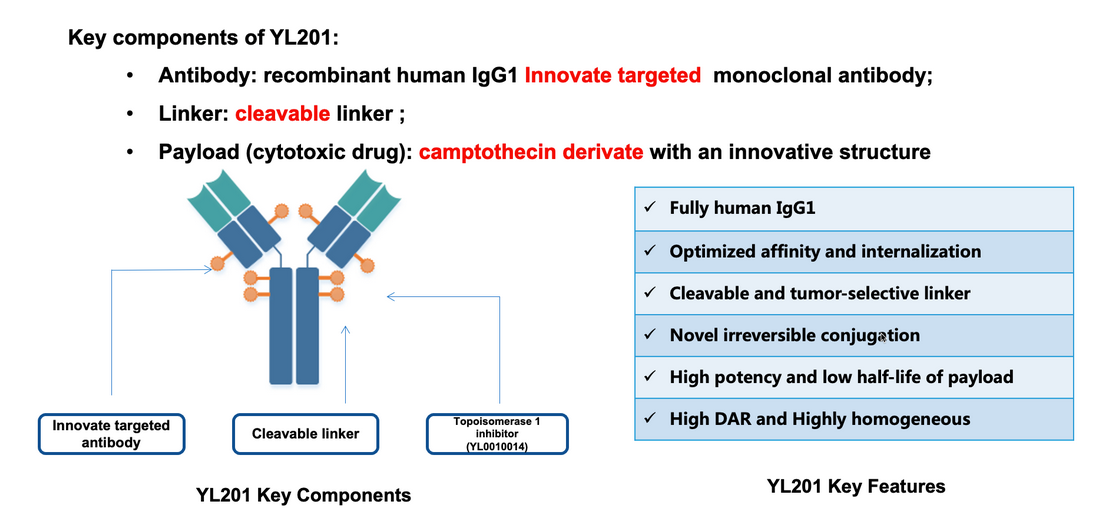
Structural Specific Binding of YL201’s Key Components
Global clinical studies of YL201 are underway for multiple advanced solid tumors. In China, YL201 has entered Phase III registration clinical trials for two indications: small cell lung cancer (SCLC) and nasopharyngeal carcinoma. Early clinical data in patients with second-line SCLC have demonstrated impressive objective response rates and survival benefits. In June 2025, the U.S. FDA granted Breakthrough Therapy Designation (BTD) to YL201 for the treatment of SCLC. Prior to this, YL201 had already obtained three Orphan Drug Designations (ODD) from the U.S. FDA, covering SCLC, nasopharyngeal carcinoma, and esophageal squamous cell carcinoma. These advancements indicate that YL201 is poised to bring new treatment hope to patients worldwide.
TMALIN® Platform: Reshaping ADC Development Paradigm with Tumor Microenvironment Cleavage Technology
MediLink Therapeutics’ TMALIN® technology platform is the core cornerstone of this global collaboration. Through its unique extracellular enzymatic cleavage mechanism in the tumor microenvironment, this platform overcomes the limitations of traditional ADC technologies: it enables the localized and specific release of payloads in tumor tissues, significantly expands the scope of antibody selection, and generates a potent bystander effect, thereby exhibiting outstanding therapeutic potential against heterogeneous tumors.
Furthermore, ADCs constructed based on the TMALIN® platform possess high circulatory stability, excellent physicochemical properties, and highly homogeneous site-specific conjugation (DAR=8), which markedly enhance the safety and development success rate of the drugs.
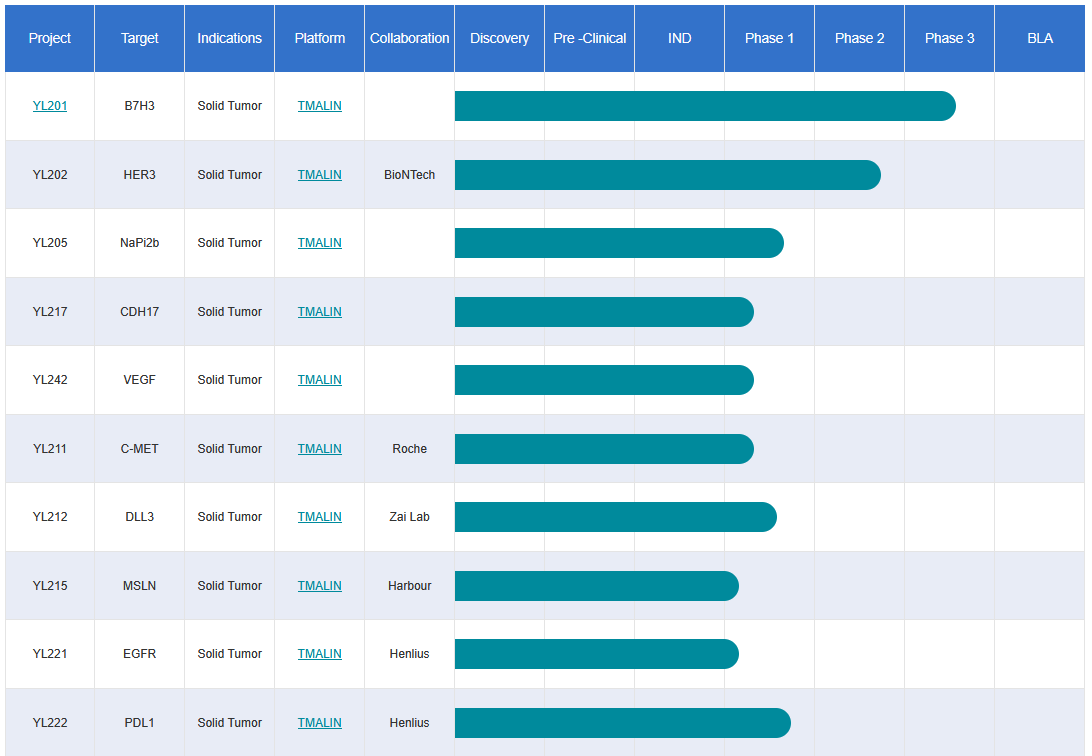
MediLink Therapeutics’ Pipeline
Leveraging its prominent technological advantages, MediLink Therapeutics’ TMALIN® platform has entered into licensing collaborations with numerous companies including Roche, BioNTech, Evopoint , Harbour BioMed, Henlius, and Zai Lab. To date, more than 10 technology or product licensing deals based on this platform have been secured, with 13 related drugs advancing to clinical research phases and 6 drugs licensed for overseas markets. This original ADC technology platform originating from China has gained high recognition from global pharmaceutical enterprises, fully demonstrating its strong international competitiveness and clinical translation potential.
Medicilion’s ADC Drug R&D Service Platform: Enabling Over 30 ADC Drugs to Gain Clinical Approval
In the field of ADC drug R&D, Medicilon boasts years of practical experience and profound technical accumulation, having established an integrated preclinical R&D service platform covering ADC payload synthesis, drug conjugation,pharmacodynamic evaluation, pharmacokinetic evaluation, and safety evaluation. As of the end of June 2025, Medicilon has successfully supported over 30 ADC drugs in obtaining clinical approval, with multiple ADC programs currently under research.
Dr. Chunlin Chen, Founder, Chairman and Chief Executive Officer of Medicilon, commented: “We congratulate MediLink Therapeutics on the successful overseas expansion of another innovative drug and the achievement of this milestone collaboration. This not only demonstrates MediLink Therapeutics’ strength in original innovation in the ADC field, but also reflects the continuous improvement of Chinese biotech companies’ capabilities in global cooperation and technology export. As a witness and enabler of the development of MediLink Therapeutics’ TMALIN® platform, we are greatly encouraged and honored. Medicilon will continue to deepen our focus on the preclinical R&D of ADCs, continuously upgrade the technical capabilities of our one-stop service platform, accelerate the R&D process of more innovative drugs, and jointly build an open, collaborative, and efficient global biopharmaceutical innovation ecosystem.”
About Medicilon
From its inception in 2004, Shanghai Medicilon Inc. (stock code: 688202.SH) has been committed to providing comprehensive R&D services to pharmaceutical companies, research institutions, and any organizations working in the preclinical space. Medicilon has built an integrated one-stop R&D platform covering drug discovery, CMC and preclinical studies, and has established service platforms in cutting-edge fields such as ADC, nucleic acids, peptides, CGT, PROTAC, and antibodies. Medicilon has established a quality system in compliance with international standards and has obtained GLP certifications from China’s NMPA, the US FDA, the EU OECD, and Japan’s PMDA, as well as AAALAC accreditation. Currently, Medicilon has nearly 80,000 square meters of R&D laboratories.
By the end of 2025, Medicilon has provided drug development services to over 2,000 clients worldwide, and has been involved in the research and development of 610+ new drugs and generic drug projects that have been approved for clinical trials with IND applications.


