



Business Inquiry
Global:
Email:marketing@medicilon.com
+1 (617) 888-9294(U.S.)
0044 7790 816 954 (Europe)
China:
Email: marketing@medicilon.com.cn
Tel: +86 (21) 5859-1500



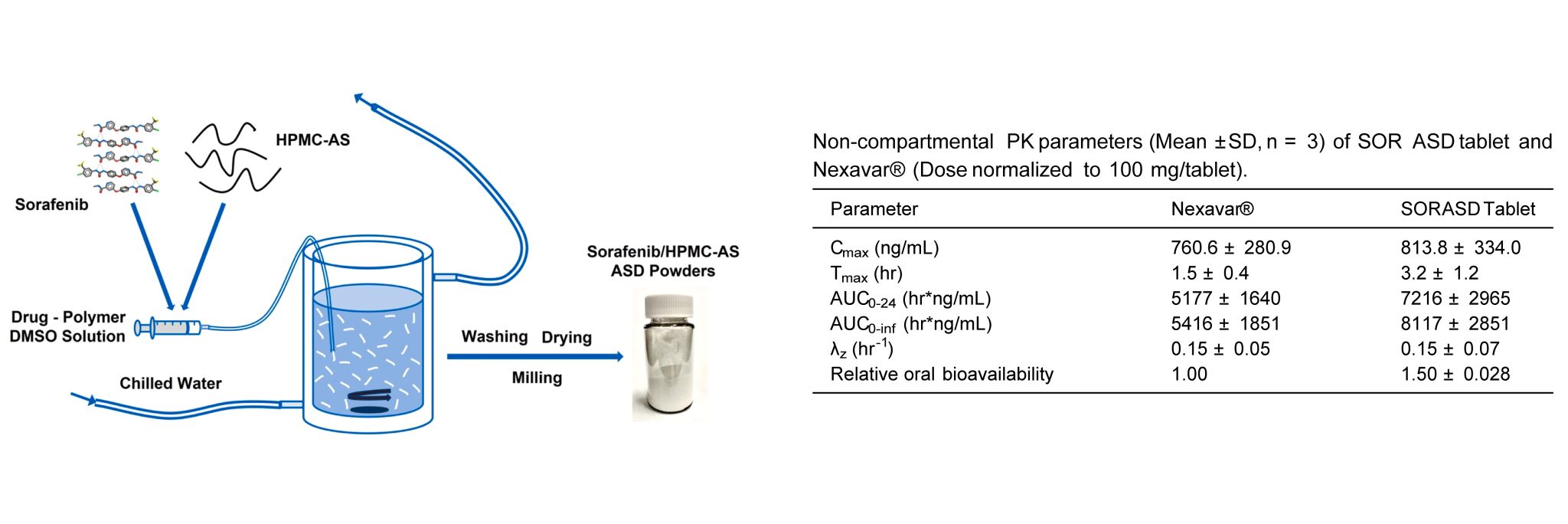 An amorphous solid dispersion (ASD) of Sorafenib (SOR) was used to develop an immediate release tablet with improved oral bioavailability. The SOR ASD tablets exhibited approximately 50% higher relative bioavailability in dogs than the marketed SOR tablet product, Nexavar®. The pharmacokinetic (PK) study of SOR ASD tablets and Nexavar® tablets was performed by Medicilon.
An amorphous solid dispersion (ASD) of Sorafenib (SOR) was used to develop an immediate release tablet with improved oral bioavailability. The SOR ASD tablets exhibited approximately 50% higher relative bioavailability in dogs than the marketed SOR tablet product, Nexavar®. The pharmacokinetic (PK) study of SOR ASD tablets and Nexavar® tablets was performed by Medicilon.
Reference:
 Relevant News
Relevant News