



Business Inquiry
Global:
Email:marketing@medicilon.com
+1(781)535-1428(U.S.)
0044 7790 816 954 (Europe)
China:
Email: marketing@medicilon.com.cn
Tel: +86 (21) 5859-1500



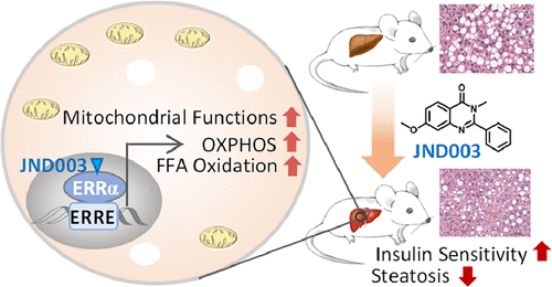
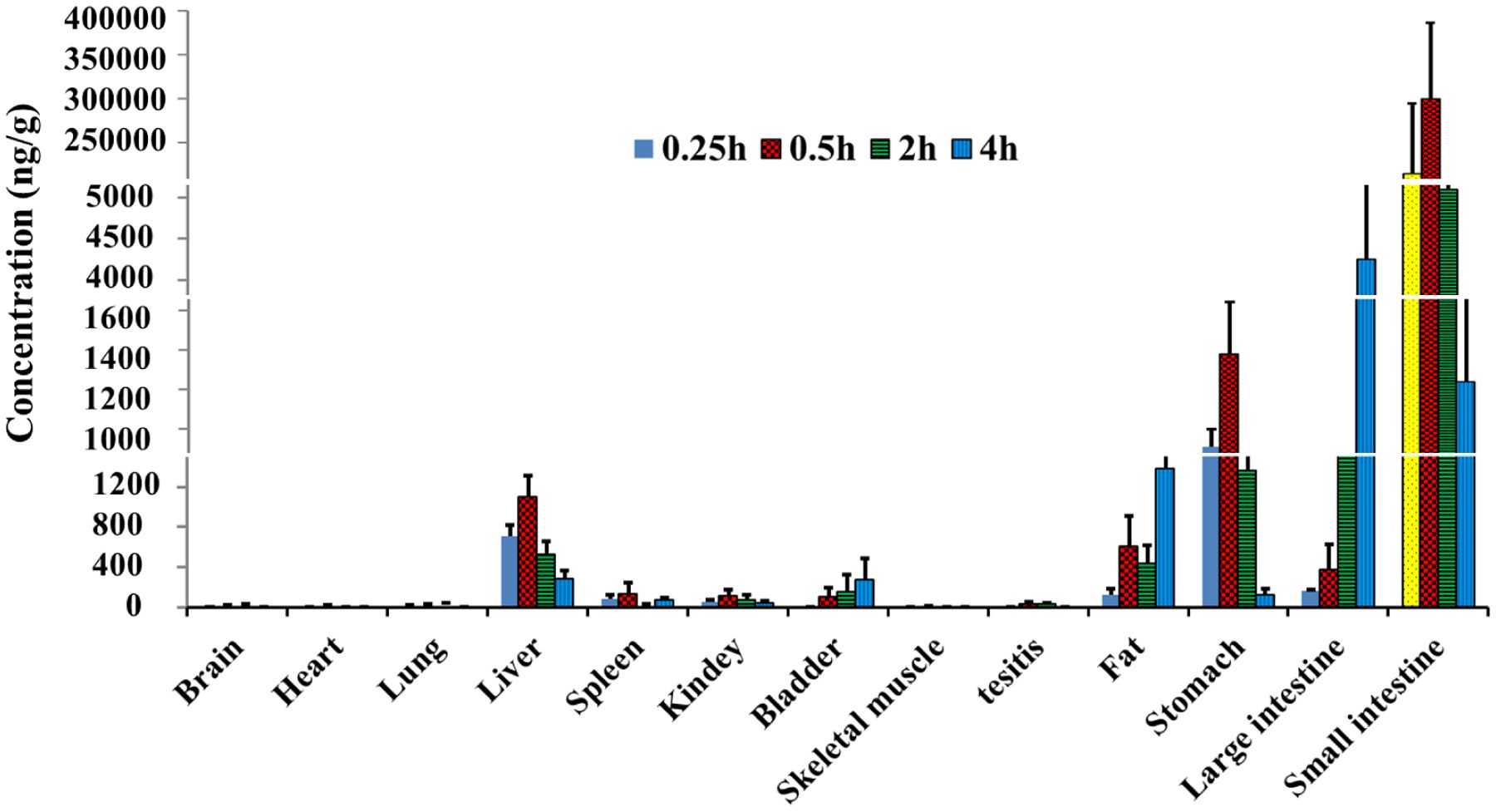
Nonalcoholic fatty liver disease (NAFLD) is one of the most prevalent forms of chronic liver diseases. Estrogen-related receptor alpha (ERRα) is a viable target for NAFLD. JND003 is a potent and selective ERRα agonist alleviating nonalcoholic fatty liver disease and insulin resistance. JND003 is orally bioavailable and exhibits high grade of distribution in liver and abdominal adipose tissues. Pharmacokinetics (PK) and Tissue Distribution Assays of JND003 were performed at Medicilon.
 Reference:
Reference:
 Relevant News
Relevant News