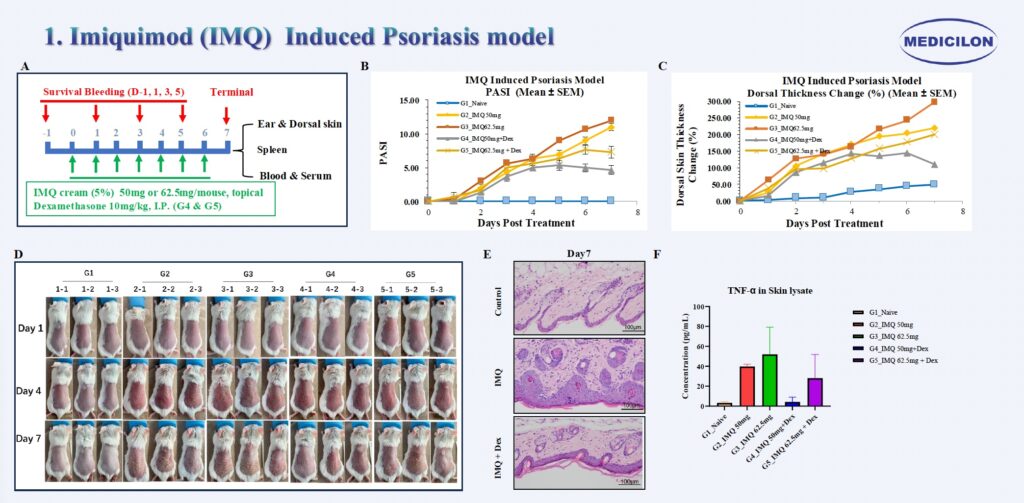
1. Imiquimod (IMQ) Induced Psoriasis model
Imiquimod (IMQ) Induced Psoriasis model. Female Balb/c mice (9 weeks old) were treated topically with imiquimod (IMQ) on the ear and dorsal skin for seven consecutive days (A). Psoriasis Area and Severity Index (PASI) (B), dorsal skin thickness change (%) (C), and dorsal skin photographs (D) were recorded daily. At the experimental endpoint, skin tissues were collected for histological evaluation. Hematoxylin and eosin (H&E) staining (E) revealed marked hyperkeratosis, epidermal hyperplasia and dense dermal leukocyte infiltration in the IMQ-treated group. Cytokine analysis withMSD further demonstrated increased expression of TNF-α in the IMQ-treated group (F).
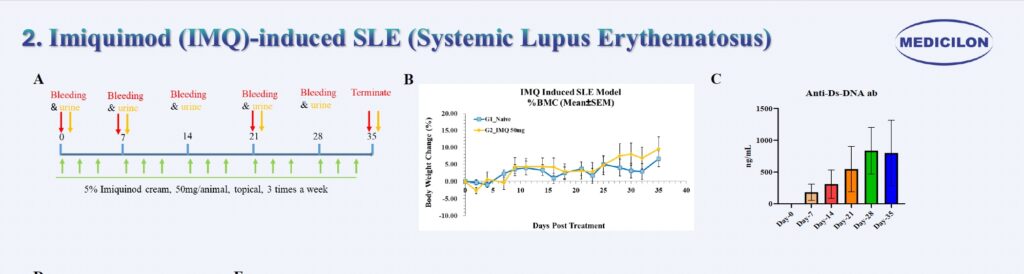
2. Imiquimod (IMQ)-induced SLE (Systemic Lupus Erythematosus
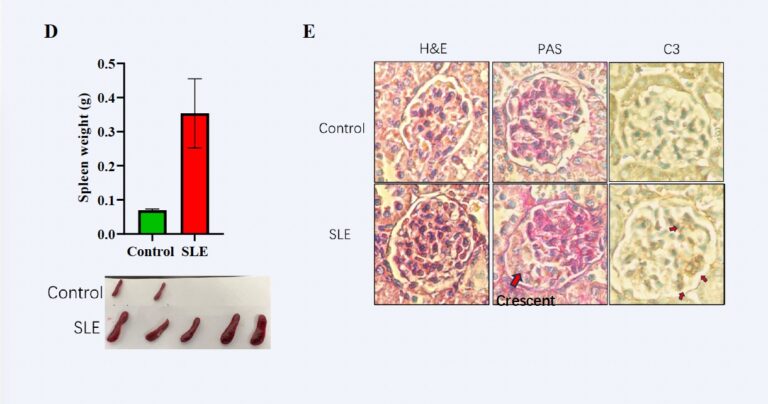
Imiquimod (IMQ)-Induced Systemic Lupus Erythematosus (SLE). Female Balb/c mice (9 weeks old) were topically treated with imiquimod (IMQ) on the ear and dorsal skin three times per week for a total duration of 5 weeks (A). Body weight was monitored thrice weekly throughout the experimental period (B). In IMQ-treated mice, anti-dsDNA antibodies became detectable as early as Day 7 and showed consistent elevation by Day 35 (C). At the study endpoint (Day 35), splenomegaly was evident, with spleen weights reaching approximately threefold higher than those of control mice (D). Histopathological evaluation (E) revealed hallmark lupus-like renal pathology, including mesangial expansion and proliferation (H&E staining), glomerular basement membrane thickening (PAS staining), and immune complex deposition (C3) detected by immunohistochemistry.
3. MC903-Induced Atopic Dermatitis Model
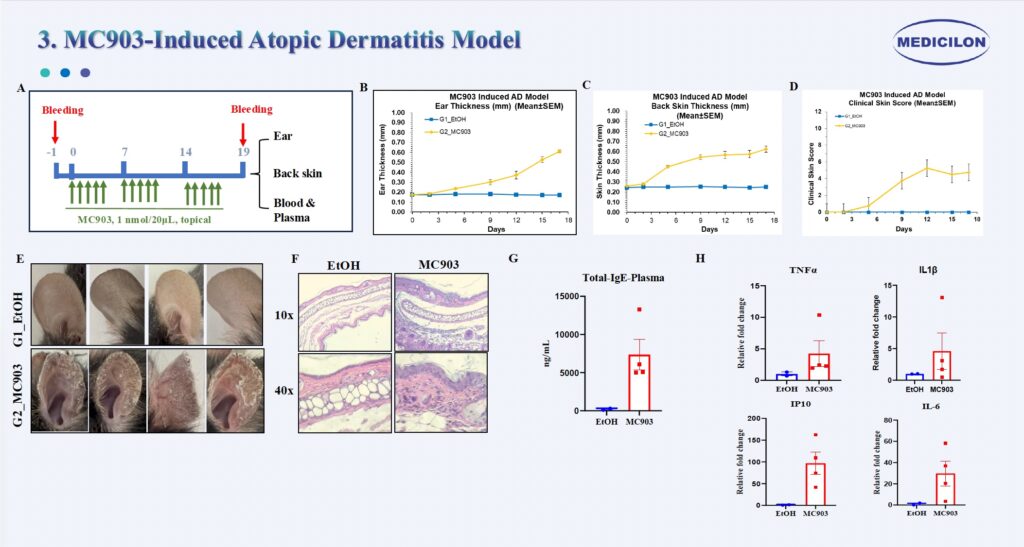
MC903-Induced Atopic Dermatitis Model. Female C57BL/6 mice (8 weeks old) were treated topically with MC903 (calcipotriol) on the ear and dorsal skin for five consecutive days per week over a 3-week period (A). Ear thickness (B), dorsal skin thickness (C), clinical skin scores (D), and ear photographs (E) were recorded twice weekly. At the experimental endpoint, skin tissues were collected for histological evaluation. Hematoxylin and eosin (H&E) staining (E) revealed marked epidermal hyperplasia and dense inflammatory cell infiltration in the MC903-treated group. Plasma total IgE levels were significantly elevated (G). Cytokine analysis further demonstrated increased expression of TNF-α, IL-1β, IP-10, and IL-6 in the MC903-treated group (F).
4. Vitiligo Mouse Model (on going)
Vitiligo Mouse Model
Category
TRP2-180 + LPS + CpG ODN 1826 Model
Pmel-1 CD8⁺T Cell Transfer Model
- Antigen Target
gp100₂₅–₃₃(KVPRNQDWL), recognized by Pmel-1 TCR
- Host Mouse
Wild-type C57BL/6, often irradiated
- T Cell Source
Transgenic CD8⁺T cells from Pmel-1 mice
- Adjuvants Used
None; IL-2 and/or peptide vaccine may be used
- Immune Activation
Adoptive transfer of effector CD8⁺T cells
- Need for Irradiation
Typically required
- Onset of Depigmentation
Day 5–7 post transfer
- Target Skin Regions
Tail, ears, muzzle, limbs
- Cellular Immunity
Monoclonal CD8⁺Pmel-1 T cells
- Advantages
Fast, robust, controlled CD8⁺T cell response
- Limitations
Requires Pmel-1 mice and irradiation; artificial TCR repertoire
- Readouts
Same, plus Pmel-1 tracking (e.g., CD90.1⁺)
- Use Cases
CD8⁺T cell function, trafficking, cytotoxicity
5. Collagen-Induced Arthritis (CIA) model
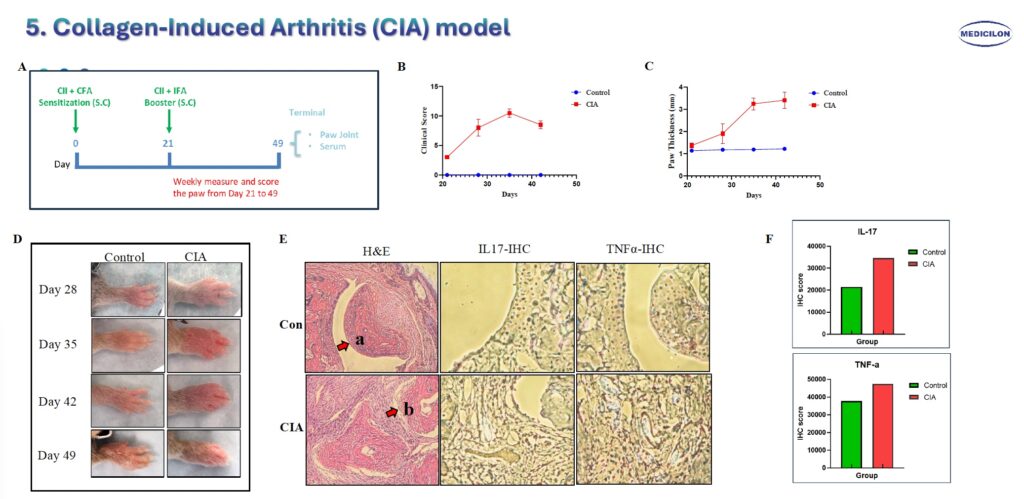
Collagen-Induced Arthritis (CIA) Model. Male DBA/1 mice (10 weeks old) were immunized with type II collagen (CII) emulsion on day 0 (200 μg) and received a booster immunization on day 21 (200 μg) (A). Clinical arthritis scores (B), paw thickness measurements (C), and representative paw images (D) were recorded weekly following the booster injection. At the experimental endpoint, bone and joint tissues were harvested for histological analysis. Hematoxylin and eosin (H&E) staining (E) demonstrated smooth articular cartilage surfaces and intact joint cavities in control mice (a), whereas CIA mice exhibited inflammatory cell infiltration and severe joint structural damage (b). Immunohistochemistry (IHC) staining further confirmed elevated expression of IL-17 and TNF-α in affected joints (E, F).
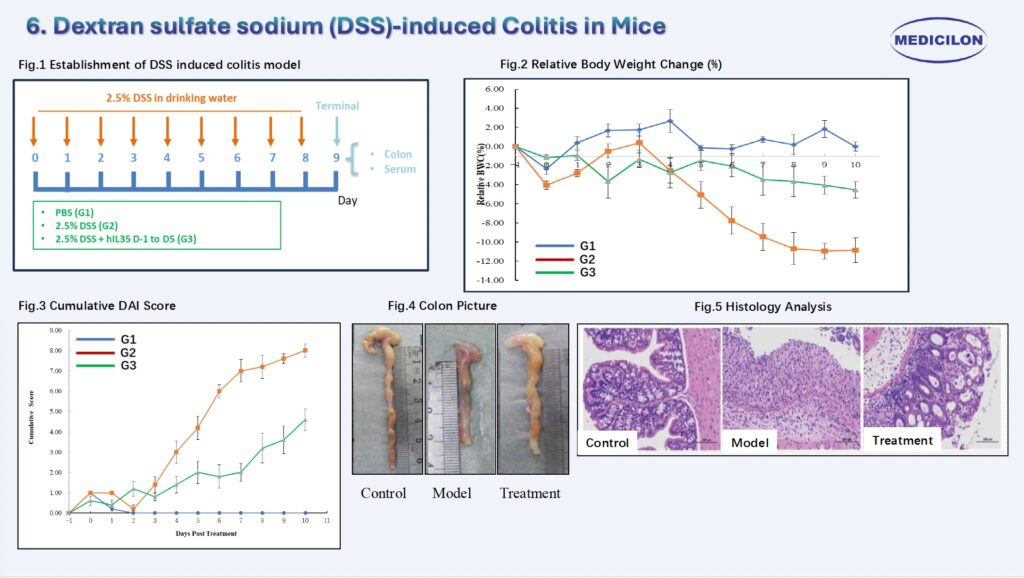
6. Dextran sulfate sodium (DSS)-induced Colitis in Mice
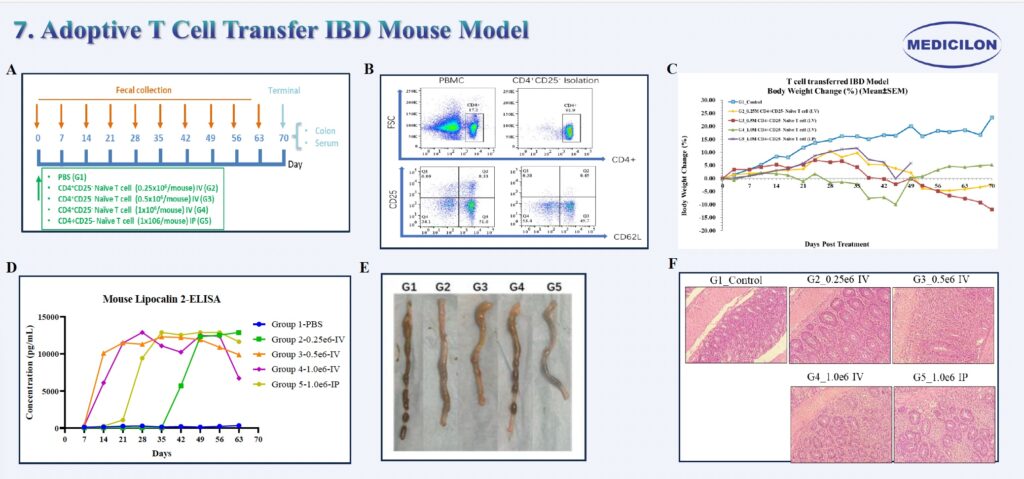
7. Adoptive T Cell Transfer IBD Mouse Model
Adoptive T Cell Transfer IBD Mouse Model. Eight-week-old female RAG2 knockout mice were intravenously injected with CD4⁺CD25⁻ naïve T cells at varying concentrations (A). The purity of CD4⁺CD25⁻ naïve T cells was confirmed by flow cytometry prior to transfer (B). Body weight was monitored three times per week throughout the experimental period (C). Fecal samples were collected weekly for measurement of lipocalin-2 levels, a biomarker of intestinal inflammation (D). At the study endpoint, colons were harvested for length measurement and histopathological analysis. The most pronounced colon shortening was observed in the G3_0.5e6_IV group (E). Hematoxylin and eosin (H&E) staining revealed crypt architectural distortion, goblet cell depletion, and marked inflammatory cell infiltration (F) .
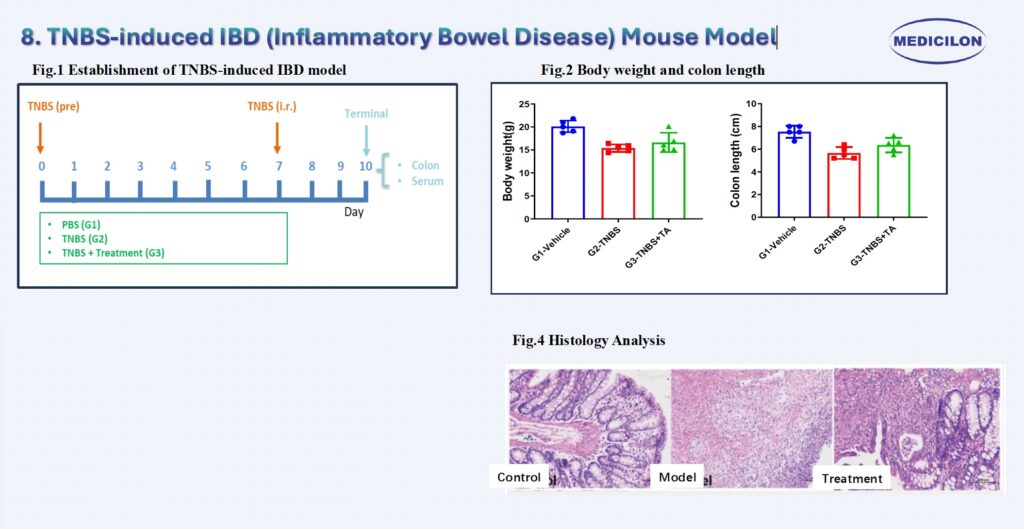
8. TNBS-induced IBD (Inflammatory Bowel Disease) Mouse Model
Feature
DSS (Dextran Sulfate Sodium)
TNBS (2,4,6-Trinitrobenzene Sulfonic Acid)
T cell Transfer (CD4⁺CD45RB^hi)
Induction Type
Chemical (epithelial toxin)
Chemical haptenation+ immune response
Immunological (adoptive transfer)
Immune Driver
Innate immune cells(macrophages, neutrophils)
Th1/Th17 cells, innate cytokines
Adaptive T cells(Th1/Th17 vs Treg imbalance)
Main Cell Types
Neutrophils, macrophages, epithelial cells
CD4⁺ T cells, macrophages, dendritic cells
CD4⁺ T cells (Th1, Th17), few B cells
Colon Segment Affected
Distal colon (continuous lesions)
Distal and sometimes proximal colon
Mainly colon (chronic)
Histopathology
Epithelial erosion, crypt loss, neutrophilic infiltration
Granulomatous transmural inflammation
Lymphocytic infiltration, crypt hyperplasia
Time Course
Acute (5–10 days)
Acute (3–7 days)
Chronic (4–10 weeks post-transfer)
Reproducibility
Very high
Moderate (depends on strain, dosing)
High but labor-intensive
Strain Sensitivity
C57BL/6, BALB/c
SJL, BALB/c
Rag1⁻/⁻ or SCID recipients
Key Cytokines
TNF-α, IL-1β, IL-6
IFN-γ, IL-12, IL-17
IFN-γ, IL-17, IL-10 (regulatory)
Resembles Human Disease
Ulcerative colitis
Crohn’s disease(Th1-driven)
Chronic Crohn’s-like colitis
Advantages
Simple, fast, reproducible, cost-effective
Reflects Th1/Th17 immunity, flexible dosing
Pure T cell–dependent, mechanistic precision
Limitations
Not immune-driven, poor chronicity
Variable severity, chemical toxicity
Long setup, requires immunodeficient mice
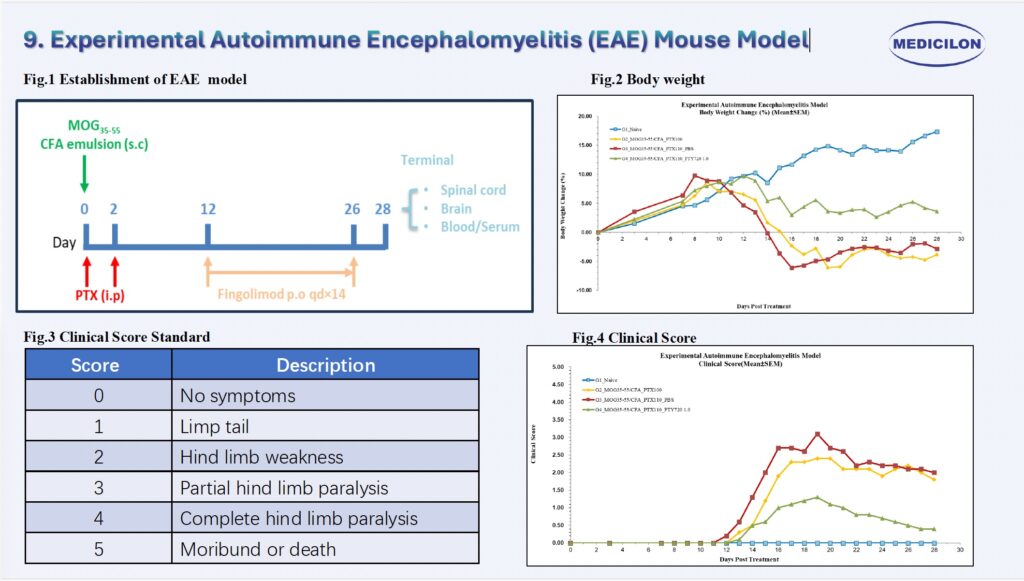
9. Experimental Autoimmune Encephalomyelitis (EAE) Mouse Model
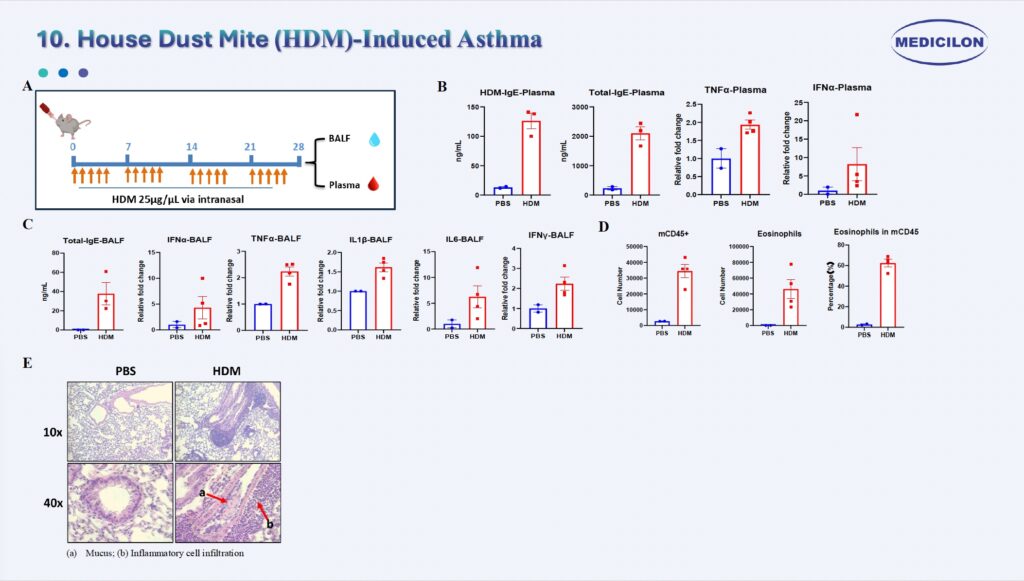
10. House Dust Mite (HDM)-Induced Asthma
House DustMite (HDM)-Induced Asthma. Female C57BL/6 mice (8 weeks old) were intranasally exposed to house dust mite (HDM) extract for five consecutive days per week over a 4-week period (A). In the HDM-treated group, total IgE levels were significantly elevated in both plasma and bronchoalveolar lavage fluid (BALF) (B, C). Cytokine profiling revealed increased systemic (plasma) and local (BALF) expression of TNF-α and IFN-α, along with locally upregulated IL-1β, IL-6, and IFN-γ (B, C). Histopathological evaluation with hematoxylin and eosin (H&E) staining (E) demonstrated pronounced mucus production (a) and dense inflammatory cell infiltration (b) in lung tissues of HDM-exposed mice. Flow cytometric analysis confirmed significant eosinophil infiltration in BALF (D), consistent with hallmark features of allergic airway inflammation.
11. Ovalbumin (OVA)-induced Asthma
Ovalbumin (OVA)-Induced Asthma. Female BALB/c mice (8 weeks old) were sensitized via intraperitoneal injection of ovalbumin (OVA) on Days 0, 7, and 14, followed by intranasal OVA challenge for seven consecutive days starting on Day 20 (A). Body weight was monitored three times per week throughout the study (B). In the OVA-treated group, total IgE levels were markedly elevated in both plasma and bronchoalveolar lavage fluid (BALF), whereas treatment with IL-35 or dexamethasone significantly suppressed IgE production (C). Cytokine profiling revealed increased systemic (plasma) and local (BALF) levels of TNF-α, IFN-γ, and IL-1β in OVA-exposed mice, which were substantially reduced following IL-35 or dexamethasone treatment (D, E). Histopathological examination using hematoxylin and eosin (H&E) staining (G) demonstrated pronounced mucus hypersecretion (a) and dense inflammatory cell infiltration (b) in the lung tissues of OVA-challenged mice. Flow cytometric analysis further confirmed significant eosinophil infiltration in BALF (F), consistent with hallmark features of allergic airway inflammation.
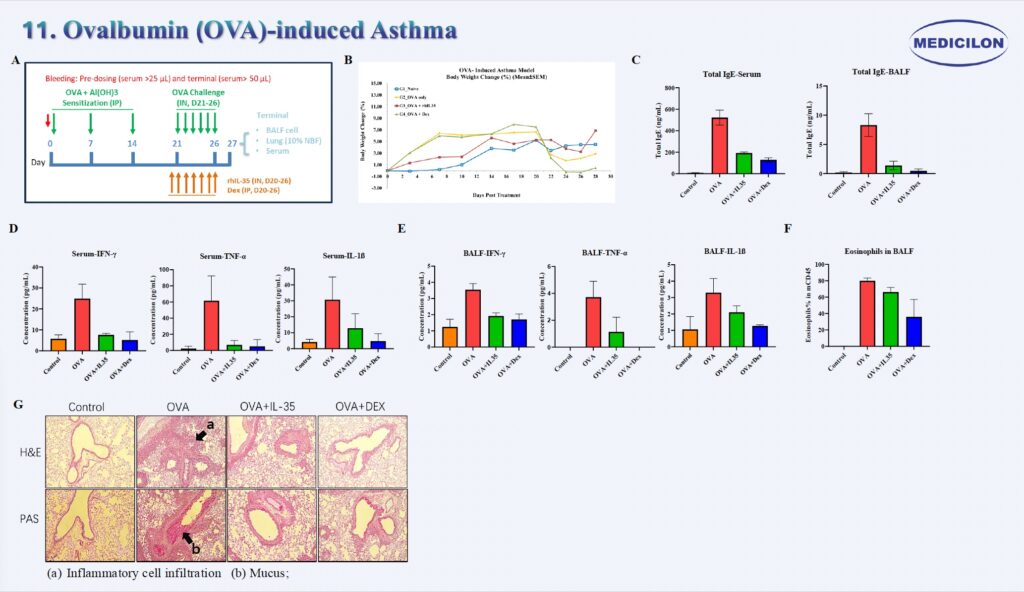
Ovalbumin (OVA)-induced Asthma
Feature
HDM-Induced Asthma Model
OVA-Induced Asthma Model
- Allergen type
Purified protein antigen (chicken egg ovalbumin)
- Relevance to human asthma
Moderate–low—artificial antigen, not normally encountered by humans
- Immune mechanism
Primarily Th2 adaptive response
- Adjuvant requirement
Requires adjuvant (usually alum) for sensitization
- Sensitization route
Intraperitoneal (sensitization) + intranasal (challenge)
- Cytokine profile
IL-4, IL-5, IL-13
- Pathology
Strong eosinophilic inflammation and airway hyperreactivity, less epithelial damage
- Chronicity
Mostly acute inflammation, less remodeling
- Innate immune involvement
Weak (mainly adaptive Th2)
- Model duration
3–4 weeks
- Reproducibility
Very high (simple and consistent)
- Application
Mechanistic studies of Th2-driven allergy and drug screening
12. Bleomycin-induced Pulmonary Fibrosis Model

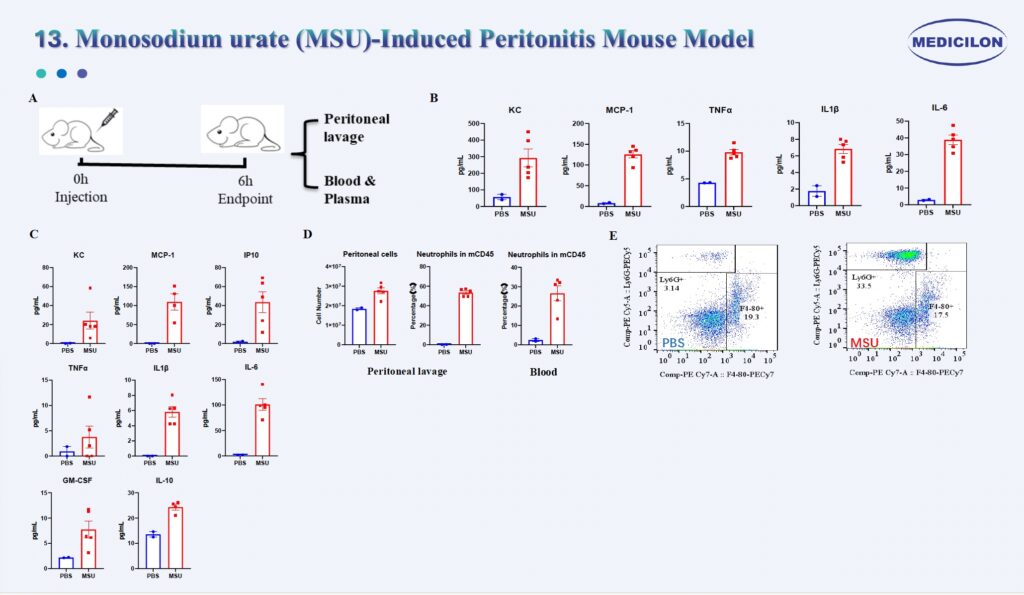
13. Monosodium urate (MSU)-Induced Peritonitis MouseModel
Monosodium urate (MSU)-Induced Peritonitis Mouse Model. Female C57BL/6 mice (8 weeks old) received an intraperitoneal injection of monosodium urate (MSU). Peritoneal lavage fluid and plasma were collected 6 hours post-injection (A). Cytokine analysis demonstrated elevated levels of pro-inflammatory cytokines (TNF-α, IL-1β, and IL-6) and chemokines (KC and MCP-1) both locally (peritoneal lavage) and systemically (plasma) in the MSU-treated group (B, C). Flow cytometric analysis confirmed a marked influx of neutrophils into the peritoneal cavity 6 hours after MSU administration (D, E).
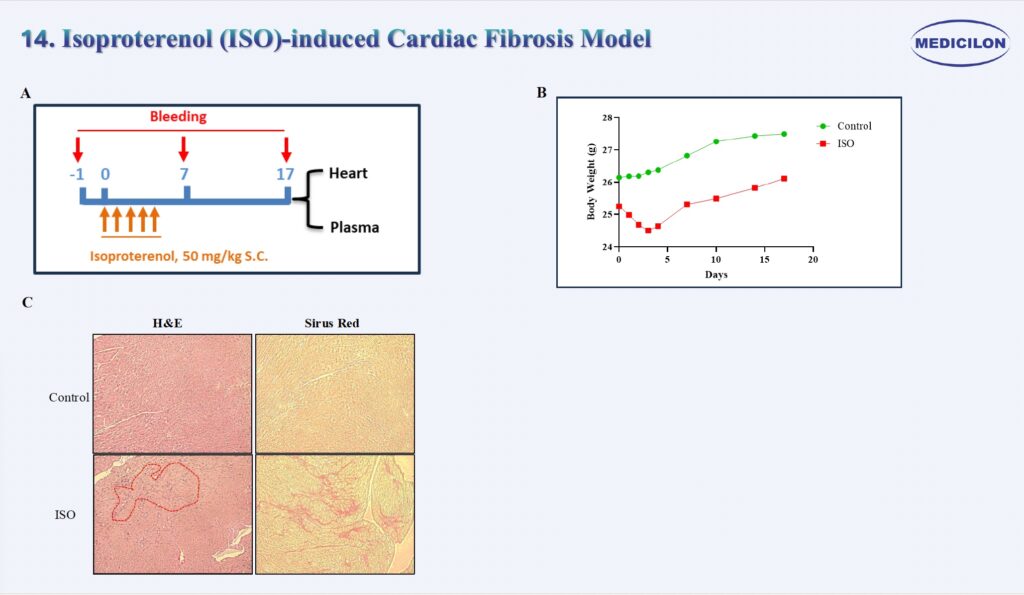
14. Isoproterenol (ISO)-induced Cardiac Fibrosis Model
Isoproterenol (ISO)-induced Cardiac Fibrosis Mouse Model. Male 129S1/SvImJ mice (12 weeks old) were subcutaneously administered Isoproterenol (ISO) for five consecutive days (A). Body weight was monitored daily during the treatment period and subsequently three times per week throughout the study (B). At the study endpoint, hearts were harvested for histological analysis. Picrosirius red staining revealed collagen fiber deposition within the myocardium, indicative of fibrotic remodeling.