Home » Advanced technology platforms » NAMs Platform
New Approach Methodologies (NAMs) Drug R&D Service Platform
New Approach Methodologies
Overview
What Are New Approach Methodologies?
New Approach Methodologies (NAMs) are a broad set of in vitro, ex vivo, and in silico approaches used to generate human-relevant evidence for drug safety and efficacy questions.
In practice, NAMs preclinical strategies are most valuable when applied in a defined context of use and integrated into a weight-of-evidence strategy to inform key development decisions.
Earlier Identification
Identify safety liabilities using human-relevant testing systems
Mechanistic Insight
Gain hypothesis-driven understanding for smarter risk assessment
Faster Iteration
Accelerate candidate optimization cycles with rapid screening
Regulatory-Ready
Build complementary data that strengthens IND narratives
Specialized Platforms
Three Integrated Pillars of NAMs Excellence
- Protein structure prediction & simulation
- Binding site identification
- Molecular optimization workflows
- Customized project databases
- Cardio, hepato & nephrotoxicity panels
- Genotoxicity & phototoxicity testing
- Immune profiling & CRA
- Organoid drug testing
- Human PK prediction
- Dose selection rationale
- Drug-drug interaction assessment
- Translational planning support
Core Capabilities
Comprehensive NAMs Toxicology Modules
hERG patch clamp;
Nav1.5/Cav1.2 assays;
iPSC cardiomyocytes (2D/3D)
Liver organoids;
HepG2/primary hepatocyte systems;
mitochondrial assays
MDCK, HEK293, Vero, HK-2;
primary cynomolgus/rat/ monkey kidney cells
Cytokine release assays;
T/B cell activation;
MLR;
PBMC-PDXO co-culture
Mini Ames, enhanced Ames;
chromosomal aberration;
micronucleus assays
12 tumor types, 43+ PDXO models;
CTG, LDH;
RNA-seq;
IHC readouts
In vitro 3T3 neutral red uptake assay for photosafety assessment
In vitro hemolysis assay for injectable formulation risk assessment
Case Studies
Representative Case Studies
Hepatotoxicity
Case 1: 2D v.s. 3D Hepatocyte Model for Drug-Induced Toxicity
A 2D HepG2 culture and 3D HepG2 spheroid system demonstrated how 3D models support longer-term, repetitive exposure designs for assessing acetaminophen-induced toxicity with enhanced mechanistic insight.
Immunotoxicity
Case 2: Cytokine Release Assay for Immune Activation Risk
CRA using fresh human PBMCs (multiple donors) with multi-cytokine panels identified potential immune activation risk early in biologics development, enabling dose optimization before clinical entry.
Platform Safety
Case 3: Platelet Viability & Activation Assessment
Fresh human platelets were used for toxicity (CTG readout) and activation assessment (CD62P flow cytometry), supporting comprehensive risk assessment for platelet liabilities.
Multi-omics
Case 4: Off-Target Assessment for Oligonucleotides
Transcriptomics (RNA-seq) evaluated potential off-target effects of oligonucleotides in cell systems, while proteomics workflows supported off-target profiling for PROTACs under different conditions.
Why NAMs
The Strategic Value of NAMs in Your Program
Identify potential safety liabilities before committing to costly in vivo studies
Non-GMP pilot batch production for early scale-up data.
Manufacturing of batches specifically for toxicology studies.
CMC documentation for China (NMPA) and U.S. (FDA) submissions.
Specialized Platforms
AI-Assisted Drug Discovery (Target → PCC)
The Role of NAMs in Modern Preclinical Strategy
- Earlier identification of safety liabilities using human-relevant testing
- Mechanistic insight for hypothesis-driven risk assessment
- Faster iteration cycles for candidate optimization
- Complementary data that strengthen regulatory narratives
These benefits make NAMs toxicology an increasingly important component of NAMs preclinical development programs.
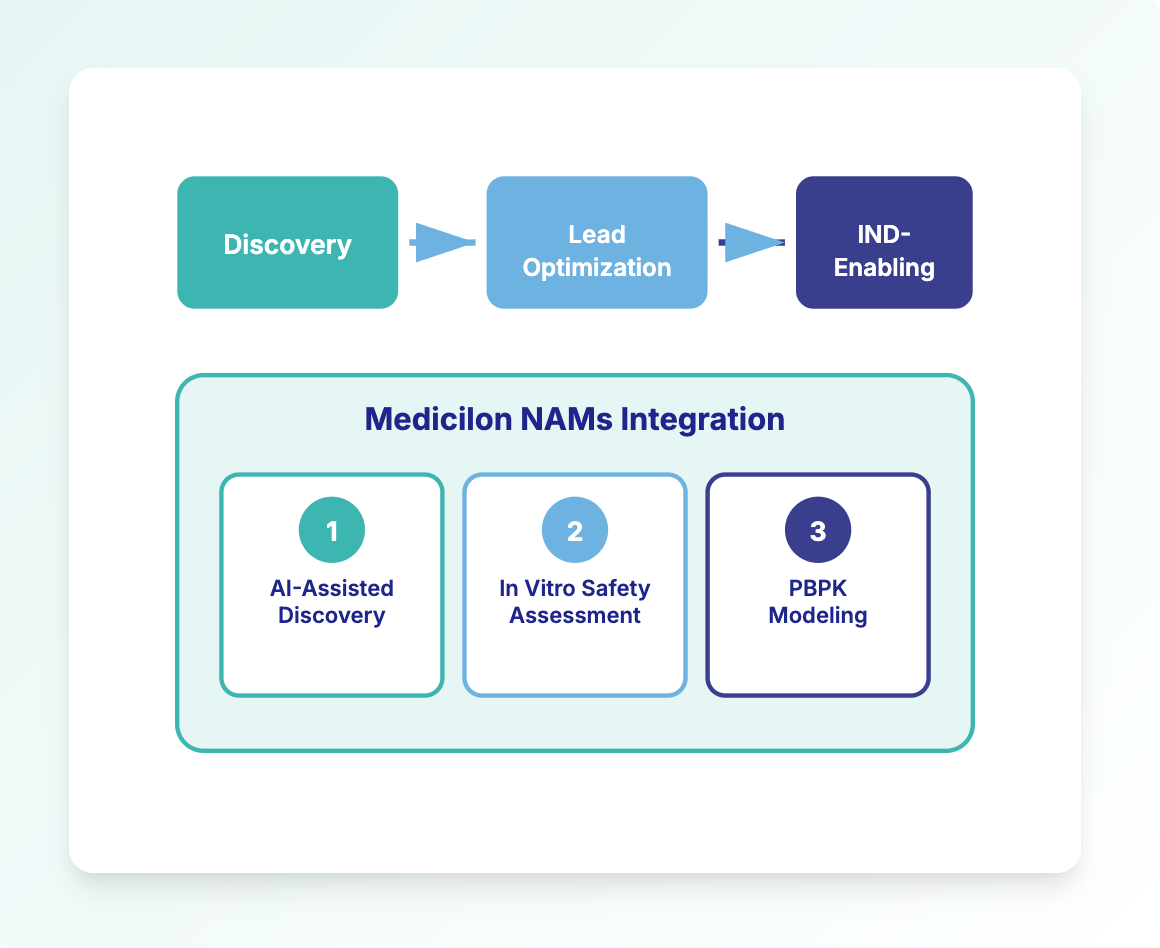
How Medicilon Applies NAMs
Medicilon supports NAMs adoption across three pillars:
- AI-assisted drug discovery support (target-to-hit, hit-to-lead, lead-to-PCC)
- In vitro safety & pharmacology profiling with organ-relevant models and mechanistic endpoints
- Dry-wet integration: linking in vitro results with modeling (PBPK/QSP/QST) to inform human translation and dosing
In Vitro Safety Assessment & Mechanistic Profiling
As an experienced in vitro toxicology CRO, Medicilon offers NAMs toxicology modules that include:
• Cytotoxicity and mechanistic assays
• Cardio-, hepato-, and nephrotoxicity panels
• Genotoxicity and phototoxicity testing
• Immune profiling and immune activation assay CRO services, including CRA
• Organoid drug testing for oncology and immuno-oncology
These human-relevant safety testing platforms support decision-making and candidate prioritization within NAMs preclinical strategies.
Core capability modules
Study
Experiment
- In vitro pharmacology profiling
- GPCRs
- Ion channel
- Enzyme
- Nuclear receptor
- Transporter
- Cytotoxicity
- CellTiter Glo/LDH/MTT/CCK-8
- Apoptosis/Necrosis/Ferroptosis
- HepG2, HUVEC, HEK293, SH-SY5Y, etc
- Primary hepatocyte, PBMC, red blood cells, platelet, etc
- Mitochondrial toxicity
- ROS assay
- Mitochondrial membrane potential assay
- Cardiotoxicity
- hERG patch clamp and radio ligand binding assays
- 5 and Cav1.2 radio ligand binding assays
- iPSCs derived cardiomyocytes-2D/3D cell toxicity
- Hepatotoxicity
- Liver organoid viability
- HepG2/primary hepatocyte mitostress-seahorse assay
- Mitochondrial membrane potential
- ROS assay
- Nephrotoxicity
- MDCK, HEK293, Vero, HK-2 cytotoxicity assay
- Primary cynomolgus, rat or monkey kidney cell cytotoxicity assay
- Genotoxicity
- Mini Ames test
- Ames test
- Enhanced Ames test
- In vitro chromosomal aberration test
- In vitro micronucleus assay
- Organoids toxicity
- Established 12 tumor types, 43 tumor organoid models.
- Toxicity testing – CTG, LDH
- Transcriptomics – RNAseq
- Immunohistochemistry
- Immunotoxicity
- Human whole blood/PBMC cytokine release assay
- T cell proliferation/activation
- B-cell proliferation/activation
- MLR(Mixed lymphocyte Reaction)
- PBMC-PDXO co-culture
- Phototoxicity
- In vitro 3T3 neutral red uptake assay
- Formulation safety
- In vitro hemolysis assay
- Weight of evidence (WoE) analysis
- Carcinogenicity
- Juvenile animal safety
- Reproductive toxicity
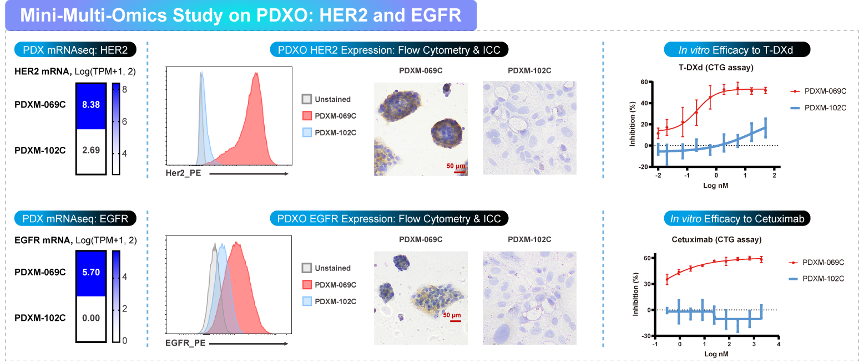
Organoid-based models for oncology and immuno-oncology
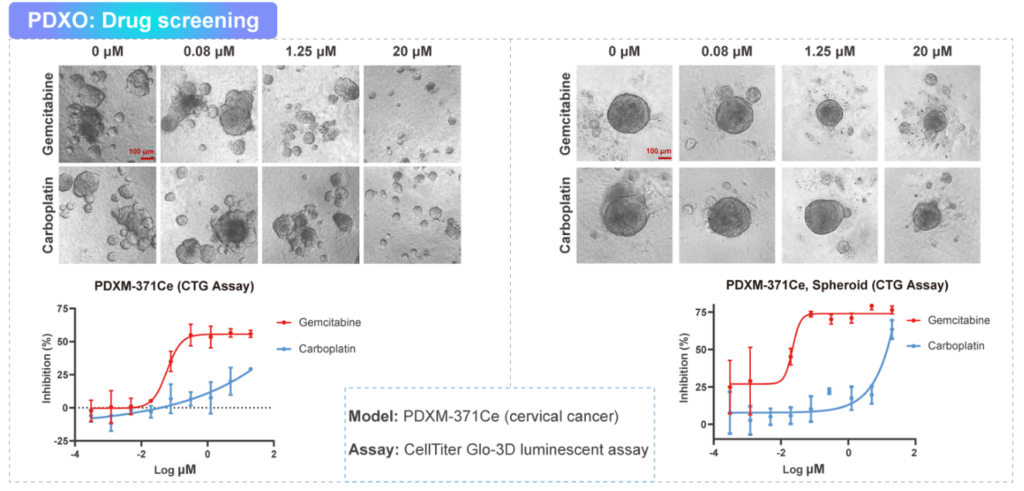
Medicilon’s PDXO platform provides patient-derived xenograft organoids (PDXOs) and related models for in vitro efficacy and mechanistic studies. PDXO models can support candidate ranking, biomarker evaluation, and co-culture studies.
PDXO drug screening with ATP-based (CTG_3D), LDH release, and imaging-based readouts
Model characterization: morphology/bright-field imaging, H&E, immunohistochemistry/IF, RNA-seq/WES as needed
PBMC–PDXO co-culture for immune cytotoxicity evaluation (e.g., infiltration and killing readouts over time)
Platform Drivern
PBPK Modeling for Human Translation
PBPK modeling services are widely used to support human PK prediction, dose selection, and DDI assessment. Medicilon integrates PBPK modeling with in vitro data to support NAMs IND support and translational planning.
This approach strengthens the scientific rationale behind NAMs toxicology and human-relevant safety testing.
- Structural formula, molecular weight, pKa, LogP/LogD
- Solubility (including SGF/FaSSIF/FeSSIF), permeability
- Plasma protein binding and blood-to-plasma ratio
- In vitro metabolic stability (microsomes/hepatocytes/recombinant enzymes) and transporter data
- Preclinical IV and extravascular PK; mass balance data where available
For IND-bound programs, NAMs preclinical data are most valuable when framed within a regulatory context
Medicilon supports: Context-of-use definition; Fit-for-purpose NAM module selection; Weight-of-evidence integration; Clear reporting to support NAMs IND-enabling packages.
When appropriately positioned, NAMs toxicology data can clarify risk assessment and improve regulatory communication.
Frequently Asked Questions
Have questions about NAMs Studies?
What are New Approach Methodologies (NAMs) in drug development?
New Approach Methodologies are a set of in vitro, ex vivo, and in silico approaches used to generate human-relevant evidence for drug safety and efficacy. In drug development, NAMs preclinical strategies support decision-making from discovery through IND preparation by providing mechanistic and translational insight.
How can NAMs support my IND-enabling strategy?
NAMs IND support focuses on applying New Approach Methodologies within a defined context of use to: 1) Identify safety liabilities earlier; 2) Generate mechanistic insight; 3) Provide complementary data for weight-of-evidence packages. Medicilon helps integrate NAMs toxicology into regulator-aware narratives that support IND-enabling planning.
Which types of assays and models does Medicilon offer under its NAMs platform?
Medicilon’s NAMs preclinical platform includes: • Cytotoxicity and mechanistic assays • Cardio-, hepato- and nephrotoxicity panels • Genotoxicity and phototoxicity testing • Immunotoxicity and cytokine release assays • Organoid drug testing for oncology and immuno-oncology • Formulation safety assays These services position Medicilon as an experienced in vitro toxicology CRO delivering human-relevant safety testing.
Do you offer organoid and PDXO models for oncology programs?
Yes. Medicilon provides organoid drug testing and a PDXO platform covering multiple tumor types for in vitro efficacy and mechanistic studies. PBMC–PDXO co-culture models can also support immune cytotoxicity evaluation in oncology programs.
How does PBPK modeling fit into NAMs?
Medicilon integrates PBPK modeling services with in vitro data to support: • Human PK prediction • Dose selection • Drug–drug interaction assessment PBPK modeling strengthens translational planning and reinforces the scientific rationale behind NAMs toxicology and human-relevant safety testing.
Can Medicilon help define where NAMs fit in my specific program?
Yes. Medicilon supports: • Context-of-use definition • Fit-for-purpose NAM module selection • Weight-of-evidence integration This ensures NAMs preclinical strategies and NAMs IND support are aligned with each program’s modality, risk profile, and regulatory objectives. .
What therapeutic modalities can benefit from Medicilon’s NAMs capabilities?
Multiple modalities can benefit from New Approach Methodologies, including: • Small molecules • Biologics • Oligonucleotide therapies • PROTACs • Complex or emerging modalities Sponsors use Medicilon’s NAMs toxicology, immune activation assay CRO services, organoid drug testing, and PBPK modeling services to inform candidate selection and IND-enabling plans.
When is the best time to engage Medicilon on NAMs?
Sponsors typically engage Medicilon during lead optimization through pre-IND stages. Early integration of NAMs preclinical modules helps: • De-risk candidates • Refine dosing strategies • Build regulator-ready data packages This supports stronger NAMs IND support planning before formal toxicology.
How do I get started with a NAMs project at Medicilon?
You can request a NAMs consultation or schedule a scientific discussion using the contact buttons on this page. Medicilon’s team will work with you to design a New Approach Methodologies strategy aligned with your development stage and IND timeline.